Early initiation of luspatercept derives greater benefit in ESA-naive TD LR-MDS: Post-hoc analysis of the phase 3 COMMANDS trial
10 Apr 2026
Share
STUDY DESIGN
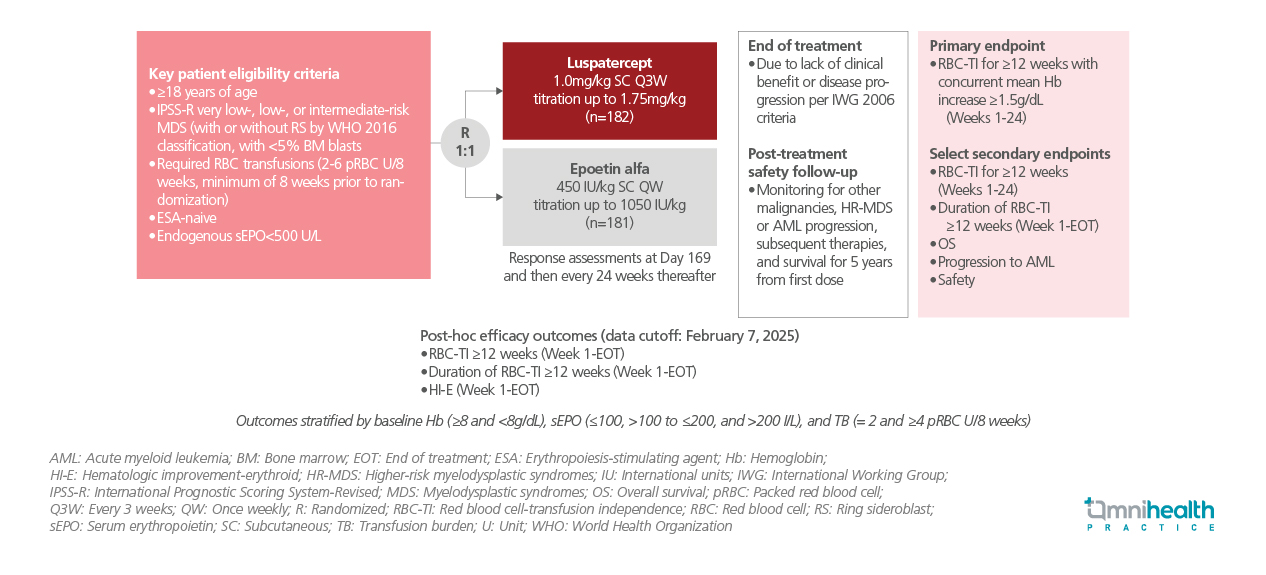
The phase 3 COMMANDS trial demonstrated the superiority of luspatercept over epoetin alfa (EA) in erythropoiesis-stimulating agent (ESA)-naive patients with transfusion-dependent (TD) lower-risk myelodysplastic syndromes (LR-MDS).1 Luspatercept achieved higher rates of red blood cell-transfusion independence (RBC-TI) and durable clinical benefit, supporting its use as the first-line treatment for anemia.1 Patient characteristics such as higher hemoglobin (Hb) levels, lower transfusion burden (TB), and lower serum erythropoietin (sEPO) levels reflect less advanced disease and are associated with greater treatment responsiveness, guiding the identification of patients most likely to benefit from treatment.1
In light of these findings, a post-hoc analysis of the COMMANDS trial was conducted to assess the impact of baseline disease severity on treatment outcomes with luspatercept compared with EA.1 Eligible patients were aged ≥18 years, ESA-naive, red blood cell-TD, and had LR-MDS, defined as Revised International Prognostic Scoring System (IPSS-R) very low-, low-, or intermediate-risk disease according to World Health Organization (WHO) classification, with <5% bone marrow blasts and endogenous sEPO levels <500U/L.1 Patients were randomized 1:1 to receive luspatercept (starting dose 1.0mg/kg, titrated up to 1.75mg/kg) administered subcutaneously every 3 weeks or EA (starting dose 450IU/kg, titrated up to 1,050IU/kg) administered subcutaneously weekly, and were followed until the end of treatment (EOT).1
The post-hoc analysis evaluated achievement and duration of RBC-TI ≥12 weeks, and erythroid hematologic improvement (HI-E) from week 1 to EOT as main efficacy outcomes, stratified by baseline Hb (<8g/dL vs. ≥8g/dL), sEPO (≤100U/L vs. >200U/L), and TB (2U vs. ≥4U of packed red blood cells over 8 weeks).1
FINDINGS
| Efficacy: |
|
‘Patients with less advanced disease, characterized by higher Hb, lower sEPO, and lower TB, achieved greater clinical benefit with luspatercept, supporting its early use in TD LR-MDS to maximize TI rates and duration.’Professor Valeria Santini
University of Florence, Florence, Italy
References
- Santini V, et al. Clinical benefit of luspatercept in erythropoiesis-stimulating agent (ESA)-naive patients with early disease characteristics and very low-, low-, or intermediate-risk myelodysplastic syndromes (LR-MDS): A post hoc analysis from the COMMANDS trial. Presented at the American Society of Hematology (ASH) Annual Meeting 2025; December 6-9, 2025.



