From nutrition to neurons: Shaping early neurodevelopment with MFGM
5 May 2026
Share
GRAPHICAL ABSTRACT
Early neurodevelopment is characterized by profound neurobiological change and high brain plasticity, during which nutrition, nurturing care, and environmental stimulation shape neural structure and function.1,2 Adverse exposures, including undernutrition, psychosocial stress, and inadequate cognitive and social input, can impair development and lead to persistent cognitive and behavioral consequences, whereas timely interventions can confer substantial and lasting benefits.1,2 Understanding the biological foundations of early brain development, the significance of critical periods in brain development, and nutritional strategies is therefore essential for optimizing neurodevelopmental outcomes.
Neurodevelopment as a public health priority
An estimated 13% of children live with developmental delays, yet only about 30% are identified before school entry, underscoring the high prevalence of these conditions.3,4 Despite this, public awareness of early brain development remains limited.5 Many parents are unaware of the critical developmental window from conception through early childhood, during which environmental and biological factors have a profound influence on lifelong outcomes.1,5,6 Strengthening awareness and early intervention is therefore essential to optimize modifiable factors and improve developmental trajectories.
First 1,000 days of life: Critical window in early brain development
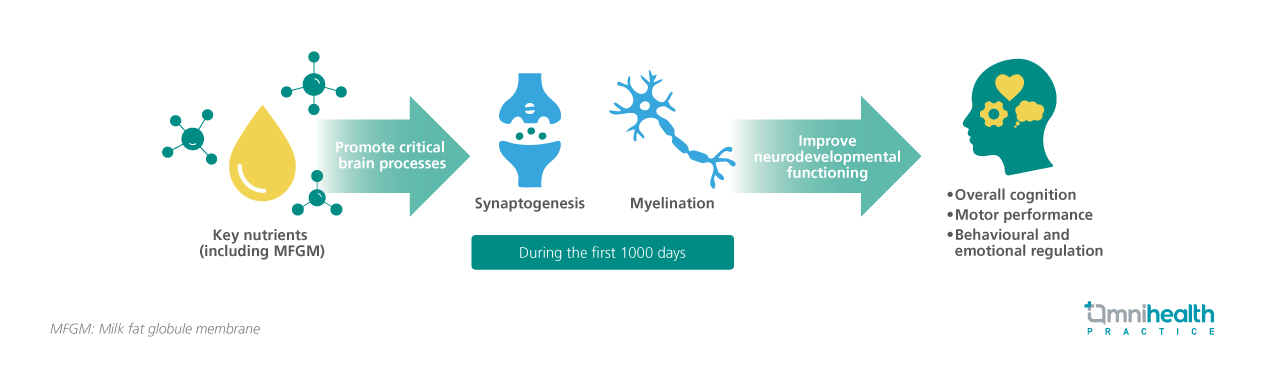
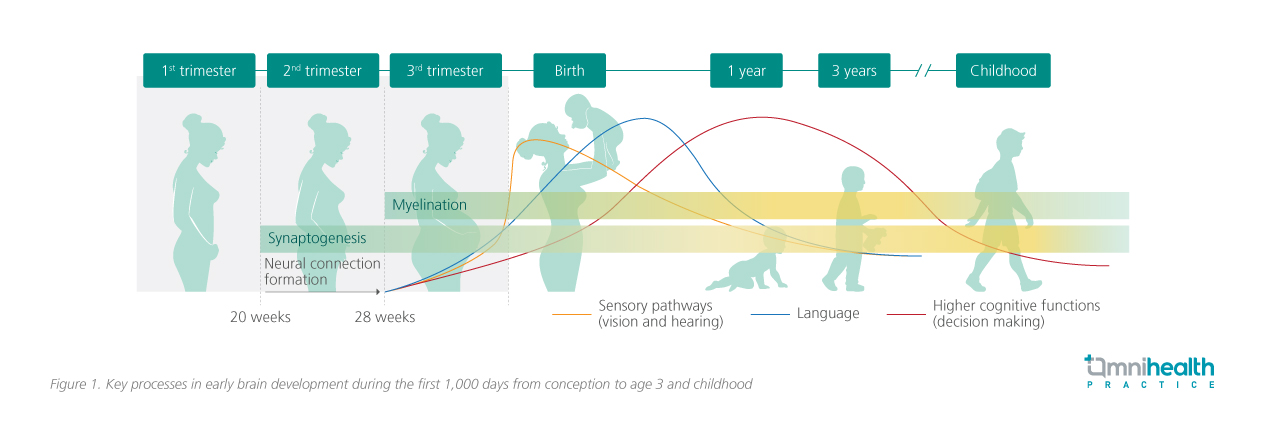
Brain development begins prenatally and accelerates during the first 1,000 days of life, a period characterized by rapid structural and functional change.6,7 During this time, foundational neural structures and systems that support core behaviors and later-emerging functions are established, including myelination and synaptogenesis (figure 1).7-11 Synaptogenesis establishes the neural circuitry for learning and memory, while increased myelination facilitates efficient information transfer, enabling cognitive and motor development.8,12 These processes occur within time-sensitive developmental windows and follow a hierarchical sequence, where more complex neural circuits depend on the successful completion of earlier stages.7 Disruption of these sequences may result in persistent functional deficits.7
 Optimal neurobehavioral development requires essential biological and environmental inputs during critical periods, as many neurodevelopmental processes cannot be fully recapitulated later.7 Infant nutrition is pivotal in shaping neurodevelopmental trajectories, with key nutrients such as choline, iron, zinc, iodine, milk fat globule membrane (MFGM), and long-chain polyunsaturated fatty acids (LC-PUFAs) supporting structural and functional maturation of the brain.6-8,12 Insufficiency during these critical windows may compromise development and contribute to long-term neurocognitive consequences beyond short-term growth.7,8
Optimal neurobehavioral development requires essential biological and environmental inputs during critical periods, as many neurodevelopmental processes cannot be fully recapitulated later.7 Infant nutrition is pivotal in shaping neurodevelopmental trajectories, with key nutrients such as choline, iron, zinc, iodine, milk fat globule membrane (MFGM), and long-chain polyunsaturated fatty acids (LC-PUFAs) supporting structural and functional maturation of the brain.6-8,12 Insufficiency during these critical windows may compromise development and contribute to long-term neurocognitive consequences beyond short-term growth.7,8
MFGM: From milk fat globule structure to neurodevelopmental function
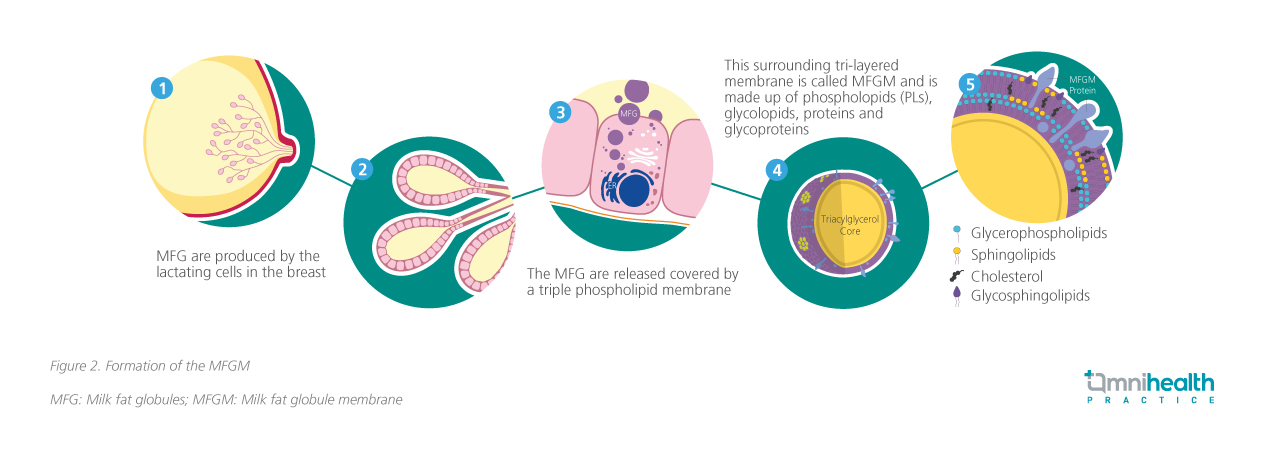
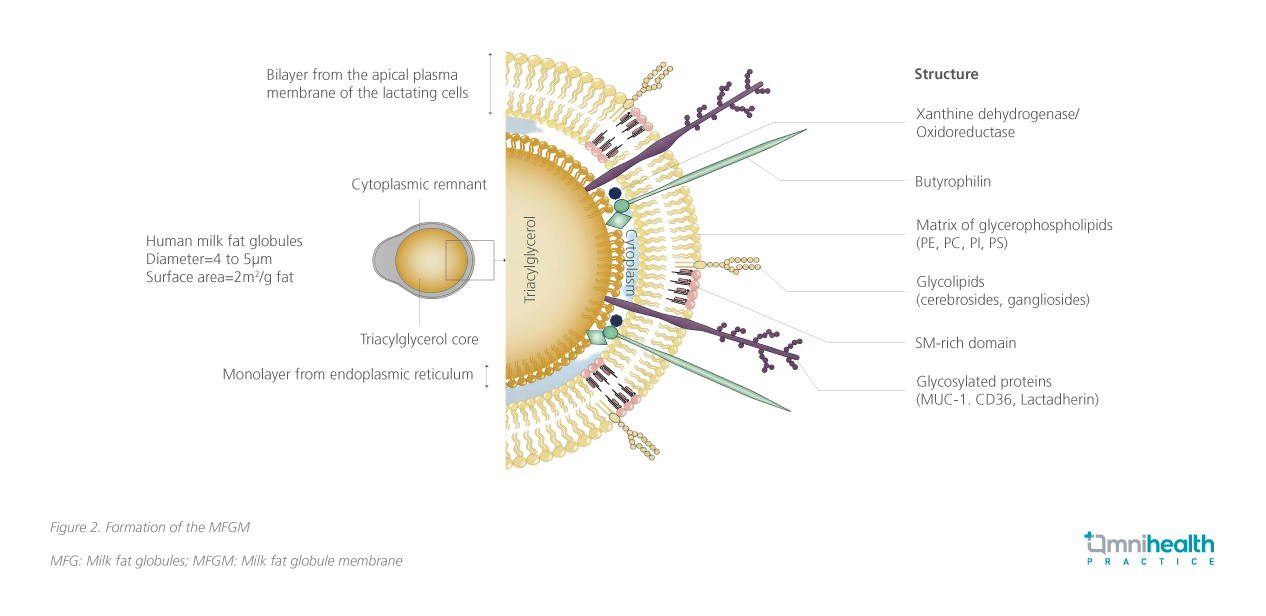
Human milk is a biologically active fluid that supports early growth, immune maturation, organ and neural development.13 Illustrated in figure 2, the lipid fraction is secreted by the lactating breast cells as milk fat globules surrounded by a specialized trilayer structure known as the MFGM.13-19 MFGM contains >150 bioactive compounds, including functional proteins, phospholipids, sphingomyelin, gangliosides, and cholesterol, which play key roles in structural and functional neurodevelopment (figure 3).14-20 Multiple MFGM components may be more impactful on neurodevelopmental outcomes than individual components of MFGM.21
Clinical evidence linking MFGM supplementation to cognitive outcomes
Mounting evidence suggests that the bioactive components of MFGM may support brain development, as well as the maturation of the gut and immune system during early life.22-30 A summary of these neurodevelopmental benefits of MFGM supplementation from multiple randomized trials is shown in table 1. Formula supplemented with MFGM supports normal growth, is well-tolerated, and safe to be consumed by infants.22,25,29,30
| Author (Year) | Population | Intervention | Comparator | Key neurodevelopmental outcomes |
| Gurnida (2012)22 | Healthy term infants | MFGM-derived complex milk lipids | Standard formula / Breastfed reference |
|
| Timby (2014)23 | Full-term infants | MFGM-supplemented formula | Standard formula / Breastfed reference |
|
| Li (2019)24 | Healthy term infants | MFGM + lactoferrin + DHA/ARA | Standard formula |
|
| Veereman-Wauters (2012)25 | Healthy preschool children | MFGM concentrate | Standard formula |
|
| Xia (2021)26 | Healthy term infants | MFGM formula | Standard formula |
|
| Nieto-Ruiz (2020, 2022)27,28 |
|
MFGM + multi-nutrient blend | Standard formula |
|
| Colombo (2023)29 | Healthy young children (age 5.5-6 years at follow-up) | MFGM + lactoferrin | Standard formula |
|
| Thongseiratch (2024)30 | Infants (0-6 years across 8 RCTs) | MFGM supplementation | Standard formula |
|
Conclusion
In conclusion, clinical evidence indicates that MFGM supplementation during the critical window of early brain development supports multiple aspects of cognitive development, including executive function, language skills, motor performance, IQ, behavioral and emotional regulation.22-30 Taken together, the overall pattern of findings is broadly consistent with those reported for breastfed infants and is more favorable than standard formula, with effects persisting into later life.22-30 These findings support that aligning infant nutrition more closely with the functional components of human milk may represent a practical strategy to enhance neurodevelopment in formula-fed infants.30
References
- Britto PR, et al. Nurturing care: promoting early childhood development. Lancet. 2017;389(10064):91-102.
- Grantham-McGregor S, et al. Developmental potential in the first 5 years for children in developing countries. Lancet. 2007;369:60-70.
- Rosenberg SA, et al. Prevalence of developmental delays and participation in early intervention services for young children. Pediatrics. 2008;121(6):e1503-e1509.
- Tomblin JB, et al. Prevalence of specific language impairment in kindergarten children. J Speech Lang Hear Res. 1997;40(6):1245- 1260.
- Royal Foundation of The Duke and Duchess of Cambridge. State of the nation: Understanding public attitudes to the early years. November 2020. Available at: https:// royalfoundation.com/wp-content/uploads/2020/11/Ipsos-MORI-SON_report_FINAL_V2.4.pdf. Accessed March 2026.
- Cusick SE, et al. The role of nutrition in brain development: The golden opportunity of the first 1000 days. J Pediatr. 2016;175:16-21.
- Schwarzenberg SJ, et al. Advocacy for improving nutrition in the first 1000 days to support childhood development and adult health. Pediatrics. 2018;141(2):e20173716.
- Derbyshire E, et al. Choline, neurological development and brain function: A systematic review focusing on the first 1000 days. Nutrients. 2020;12:1731.
- Shonkoff JP, et al. From neurons to neighborhoods: The science of early childhood development. National Academies Press (US); Available at: https://www.ncbi.nlm.nih.gov/books/NBK225557/. Accessed: March 25, 2026.
- Fleming LL, et al. Cognitive control and neural activity during human development: evidence for synaptic pruning. J Neurosci. 2024;44(26):e0373242024.
- Kostović I, et al. The development of the subplate and thalamocortical connections in the human foetal brain. Acta Paediatr. 2010;99(8):1119-1127.
- Deoni SCL, et al. Enhanced brain myelination and cognitive development in young children associated with milk fat globule membrane (MFGM) intake: A temporal cohort study. Res Sq. 2024. Preprint.
- Ballard O, et al. Human milk composition: Nutrients and bioactive factors. Pediatr Clin North Am. 2013;60(1):49-74.
- Dewettinck K, et al. Nutritional and technological aspects of milk fat globule membrane material. Int Dairy J. 2008;18:436-457.
- Oshida K, et al. Effects of dietary sphingomyelin on central nervous system myelination in developing rats. Pediatr Res. 2003;53:589-593.
- Li Z, Vance DE. Phosphatidylcholine and choline homeostasis. J Lipid Res. 2008;49:1187-1194.
- Dietschy JM, et al. Cholesterol metabolism in the central nervous system during early development and in the mature animal. J Lipid Res. 2004;45(8):1375-1397.
- Palmano K, et al. The role of gangliosides in neurodevelopment. Nutrients. 2015;7:3891-3913.
- Lopez C, et al. Human milk fat globules: polar lipid composition and in situ structural investigations revealing the heterogeneous distribution of proteins and the lateral segregation of sphingomyelin in the biological membrane. Colloids Surf B Biointerfaces. 2011; 83:29-41.
- Bode L. Human milk oligosaccharides: Every baby needs a sugar mama. Glycobiology. 2012;22(9):1147-1162.
- Brink LR, et al. Effects of milk fat globule membrane and its various components on neurologic development in a postnatal growth restriction rat model. J Nutr Biochem. 2019;69:163-171.
- Gurnida DA, et al. Association of complex lipids containing gangliosides with cognitive development of 6-month-old infants. Early Hum Dev. 2012, 88:595-601.
- Timby N, et al. Neurodevelopment, nutrition, and growth until 12 mo of age in infants fed a low-energy, low-protein formula supplemented with bovine milk fat globule membranes: A randomized controlled trial. Am J Clin Nutr. 2014;99:860-868.
- Li F, et al. Improved neurodevelopmental outcomes associated with bovine milk fat globule membrane and lactoferrin in infant formula: a randomized, controlled trial. J Pediatr. 2019;215:24-31.e8.
- Veereman-Wauters G, et al. Milk fat globule membrane (INPULSE) enriched formula milk decreases febrile episodes and may improve behavioral regulation in young children. Nutrition. 2012, 28:749-752.
- Xia Y, et al. Neurodevelopmental outcomes of healthy Chinese term infants fed infant formula enriched in bovine milk fat globule membrane for 12 months: A randomized controlled trial. Asia Pac J Clin Nutr. 2021;30(3):401-414.
- Nieto-Ruiz A, et al. Influence of a functional nutrients-enriched infant formula on language development in healthy children at four years old. Nutrients. 2020;12(2):535.
- Nieto-Ruiz A, et al. Infant formula supplemented with milk fat globule membrane, long chain polyunsaturated fatty acids, and synbiotics is associated with neurocognitive function and brain structure of healthy children aged 6 years: the COGNIS study. Front Nutr. 2022;9:820224.
- Colombo J, et al. Improved neurodevelopmental outcomes at 5.5 years of age in children who received bovine milk fat globule membrane and lactoferrin in infant formula through 12 months: A randomized controlled trial. J Pediatr. 2023;261:113483.
- Thongseiratch T, et al. Bovine milk fat globule membrane supplementation and neurocognitive development: A systematic review and meta-analysis. Nutrients. 2024;16:2374.
This is an independent editorial article, published and distributed through unrestricted educational support from the pharmaceutical community for the purpose of continuing medical education only. The views expressed in this publication reflect the experience and/or opinion of the author(s) and are not necessarily those of editors, publisher and sponsor(s). Because of rapid advances in medicine, independent verification of clinical diagnoses, medical suitability and dosage should be made before treatment prescription. The appearance of advertisement, if any, has no influence on editorial content or presentation and does not imply the endorsement of products by the publication, or its authors and editors.
